$3.575M Similasan Eye Drop Settlement Payments Going Out
By Steve Levine
Published: February 25, 2026 — Updated: February 25, 2026
Settlement Amount: $3,575,000
Status: Payments Going Out (2026)
No Proof of Purchase Required (Up to $10 Per Household)
Payment Methods: Zelle, Venmo, Direct Deposit, Prepaid Card, or Check
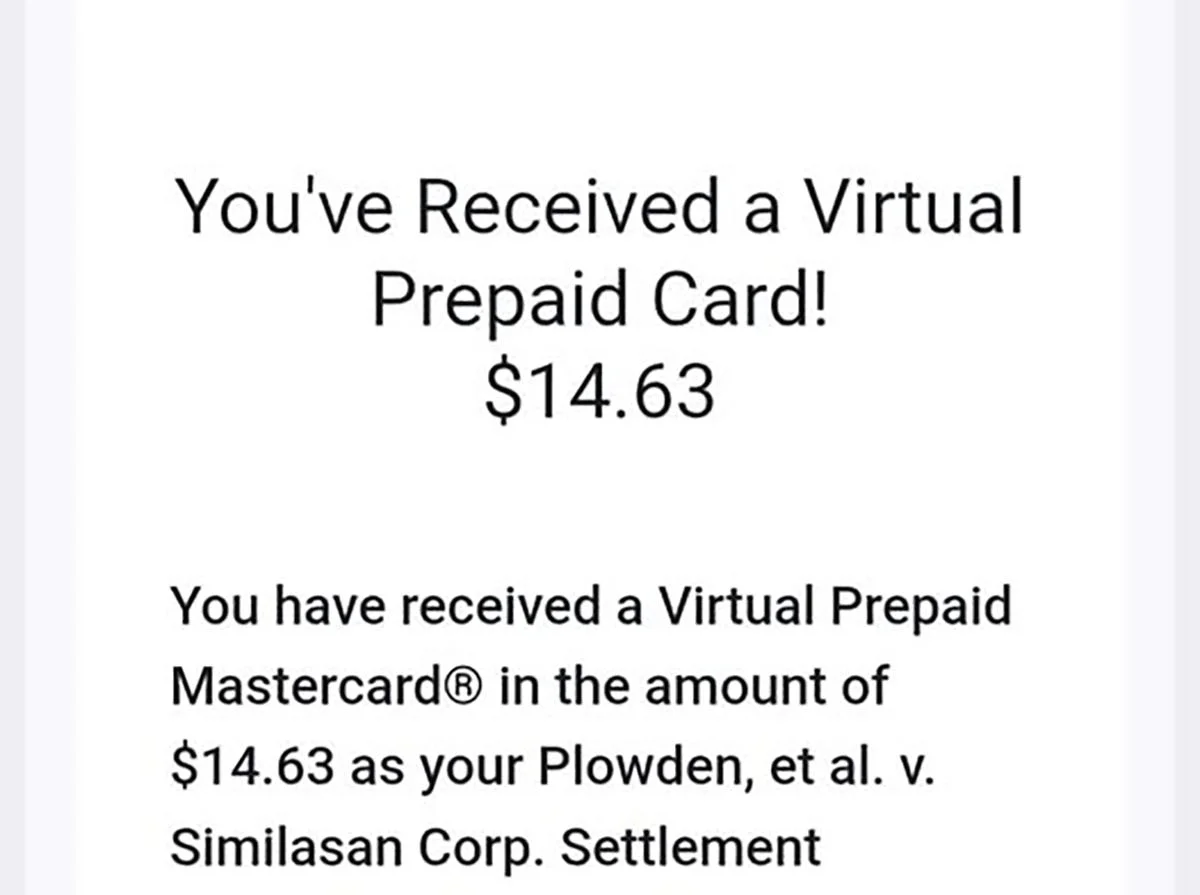
The $3.575 million Similasan eye drop class action settlement is paying out. Consumers who filed claims for Similasan, CVS, and Walgreens homeopathic eye drops are now receiving settlement payments via Zelle, Venmo, direct deposit, MasterCard virtual prepaid card, or paper check. The settlement received final court approval on July 17, 2025, and payments are being distributed after the October 15, 2025 claim deadline passed and claims processing was completed.
The lawsuit alleged that Similasan — a Swiss company that markets homeopathic health products in the United States — deceptively labeled and advertised its eye drops as “homeopathic” without proper FDA approval, misleadingly marketed the products as “sterile,” and failed to warn consumers about the risk of silver sulfate, an ingredient that can cause irreversible eye and skin discoloration. The settlement covered 16 different eye drop products sold under the Similasan, CVS, and Walgreens brand names.
No proof of purchase was required to file a claim. Consumers without receipts could claim up to $10 per household ($2.50 per product for up to 4 products). Those with proof of purchase could claim $2.50 for every product they bought with no cap. Nearly 100,000 claim forms were submitted before the deadline.
Here is the full breakdown of what happened, what your payment means, and how the settlement fund is being distributed.
See full settlement details on OpenClassActions.com
Is the Similasan Eye Drop Settlement Payment Legitimate?
Yes. The Similasan eye drop settlement payment is real and legitimate. The $3.575 million settlement in Plowden et al. v. Similasan Corp. received final approval from a federal judge in the U.S. District Court for the District of Colorado on July 17, 2025. If you filed a claim before the October 15, 2025 deadline and received a payment via Zelle, Venmo, direct deposit, MasterCard prepaid card, or check, it is a court-authorized settlement disbursement.The settlement administrator is distributing payments from the $3.575 million fund. Payments are being sent to the email address, phone number, or mailing address that claimants provided on their claim forms. If you filed a claim months ago and are now receiving an unexpected electronic payment, it is most likely this settlement.
How Much Will You Get from the Similasan Settlement?
The Similasan eye drop settlement pays $2.50 per eligible product. How much you receive depends on whether you filed with or without proof of purchase and how many products you claimed:Similasan Settlement Payment Amounts
● With Proof of Purchase: $2.50 per product, no cap — claim for every product purchased with valid receipts, frequent shopper records, or packaging
● Claims limited to one per household
● Pro rata adjustment possible: If valid claims exceed the available fund after fees and costs, payments will be reduced proportionally. If fewer claims were filed than expected, payments could be increased.
Nearly 100,000 claim forms were filed in this settlement according to court records from the final approval hearing. The direction and size of any pro rata adjustment depends on the total value of all approved claims relative to the net settlement fund after attorney fees, administration costs, and service awards are deducted.
$3.575 Million Settlement Fund Breakdown
● Administration Costs: Deducted from the fund for claims processing and payment distribution
● Service Awards: Up to $2,500 each for three named plaintiffs David Plowden, Mario Ortega, and Kamille Faye Vinluan-Jularbal ($7,500 total)
● Net Fund for Claimants: Remaining balance after all deductions
● Residual Funds: Any money remaining after all payments are distributed will be donated to The Public Justice Foundation
If every one of the approximately 100,000 claimants filed without proof of purchase and claimed the maximum $10, the total payout would be $1 million — well within the estimated net fund. This means pro rata increases are possible for this settlement, meaning you could receive more than $10 rather than less. However, claimants with proof of purchase could have claimed unlimited products, so the final calculation depends on the total value of all claims combined.
What Products Are Covered by the Similasan Eye Drop Settlement?
The settlement covers 16 homeopathic eye products manufactured, distributed, or sold by Similasan Corporation, including private-label versions sold at CVS and Walgreens. The CVS and Walgreens products were made with ingredients supplied by Similasan and are substantially similar to the Similasan-branded products.All 16 Covered Products
● Similasan Complete Eye Relief
● Similasan Allergy Eye Relief
● Similasan Kids Allergy Eye Relief
● Similasan Red Eye Relief
● Similasan Pink Eye Relief
● Similasan Kids Pink Eye Relief
● Similasan Aging Eye Relief
● Similasan Computer Eye Relief
● Similasan Stye Eye Relief
● Similasan Pink Eye Nighttime Gel
● Similasan Dry Eye Nighttime Gel
● CVS Pink Eye Drops
● Walgreens Stye Eye Drops
● Walgreens Pink Eye Drops
● Walgreens Allergy Eye Drops
To qualify, you must have purchased one or more of these products in the United States for personal use (not for resale) between September 11, 2017 and February 20, 2025. The class period covers approximately seven and a half years of purchases. Many consumers bought these products regularly at pharmacy chains, grocery stores, and online retailers, often as an alternative to conventional medicated eye drops.
What Was the Similasan Eye Drop Lawsuit About?
The lawsuit, officially titled Plowden et al. v. Similasan Corporation, was originally filed in September 2023 in the U.S. District Court for the District of Colorado. Three named plaintiffs — David Plowden, Mario Ortega, and Kamille Faye Vinluan-Jularbal — brought the case on behalf of all U.S. consumers who purchased the covered products.The complaint alleged four main violations:
First, that Similasan deceptively labeled and advertised its eye drops as “homeopathic” when the products had not secured the appropriate approvals from the FDA. The plaintiffs argued that consumers understood “homeopathic” to mean the products met certain recognized standards, when in fact they did not have FDA approval recognizing them as safe and effective.
Second, that the eye drops were not manufactured according to Current Good Manufacturing Practices (cGMP), the FDA’s quality standards for pharmaceutical production. This meant the products could not reliably ensure consistent quality, purity, or potency.
Third, that Similasan misleadingly marketed the products as “sterile” without adequate clinical validation. For eye drops applied directly to a sensitive organ, sterility claims carry significant medical weight, and the lawsuit alleged these claims were unsubstantiated.
Fourth, that the products contained silver sulfate — an ingredient the FDA does not consider safe or effective for over-the-counter ophthalmic use — without disclosing the associated risks. According to the complaint, silver sulfate can decrease night vision and cause irreversible discoloration of the eyes and skin with prolonged use. This risk was never communicated to consumers on the product labeling.
Similasan Corporation denied all allegations throughout the proceedings and entered the settlement without any admission of wrongdoing. The company asserted that its products were clearly labeled as not reviewed by the FDA. Despite the denial, Similasan agreed to pay $3.575 million to resolve the case and avoid the expense and uncertainty of continued litigation.
When Are Similasan Settlement Payments Being Sent?
The Similasan eye drop settlement received final approval on July 17, 2025. The claim deadline passed on October 15, 2025. Settlement payments are being processed and distributed in early 2026.Complete Settlement Timeline
● November 6, 2024: Settlement agreement filed with the court
● February 20, 2025: Preliminary approval granted (end of class period)
● May 16, 2025: Settlement made public
● May 27, 2025: Deadline to opt out or file objections
● July 17, 2025: Final approval hearing — final approval granted; nearly 100,000 claims filed
● October 15, 2025: Claim form deadline (passed)
● Early 2026: Settlement payments being distributed
Payments typically arrive 60 to 90 days after final approval and completion of claims processing. Electronic payments via Zelle, Venmo, and direct deposit are generally distributed faster than paper checks. If you selected a MasterCard virtual prepaid card, the card details will be sent to the email address on your claim form.
No Proof of Purchase Was Required for the Similasan Settlement
The Similasan eye drop settlement was a no proof required class action settlement for claims up to $10 per household. Consumers who could not produce receipts or purchase records could still file and receive $2.50 for each of up to 4 covered products, simply by affirming their purchases under penalty of perjury.This low barrier to entry made the settlement accessible to the vast majority of consumers who had purchased Similasan, CVS, or Walgreens homeopathic eye drops over the seven-and-a-half-year class period but had long since discarded any proof. Eye drops are a low-cost, frequent-purchase product, and virtually no one keeps receipts for them.
Consumers who did have proof of purchase — such as store receipts, frequent shopper program records, or original packaging — could claim $2.50 for every product they bought during the class period with no cap. For consumers who purchased these products regularly over many years, the no-cap provision could yield significantly higher payouts.
Valid proof of purchase included store receipts, credit card or bank statements showing the purchase, frequent shopper program records, or product packaging that documented a purchase on or before February 20, 2025.
What Is the Silver Sulfate Risk in Similasan Eye Drops?
One of the most significant allegations in the lawsuit concerned silver sulfate, an ingredient present in certain Similasan eye drop products. The complaint alleged that the FDA does not recognize silver sulfate as safe or effective for over-the-counter ophthalmic use, and that Similasan failed to disclose the associated health risks on product labeling.According to the lawsuit, prolonged use of products containing silver sulfate can lead to decreased night vision and irreversible discoloration of the eyes and skin — a condition known as argyria when it affects the skin. These are serious side effects for a product marketed as a gentle, natural remedy for common eye conditions like dry eyes, allergies, and pink eye.
Similasan denied these allegations and maintained that its products were safe. The settlement resolved the claims without any admission of wrongdoing. However, many consumers who used these products daily for years were unaware of the potential risks until the lawsuit brought them to light.
How Similasan Settlement Payments Are Being Distributed
The settlement administrator is distributing payments through five different methods, depending on what each claimant selected when filing their claim form:MasterCard Virtual Prepaid Card: A digital prepaid card number is emailed to you. You can use it for online purchases or add it to a digital wallet. No physical card is mailed.
Venmo: The payment is sent directly to your Venmo account based on the contact information on your claim form. The funds appear in your Venmo balance and can be transferred to your bank.
Direct Deposit (ACH): The payment is deposited directly into the bank account you specified on your claim form. This is typically the fastest method.
Zelle: The payment is sent to the phone number or email address associated with your Zelle account. It appears as a standard Zelle transfer in your banking app.
Paper Check: A physical check is mailed to the address on your claim form. Checks should be deposited promptly to avoid expiration.
Why Did the Similasan Settlement Attract Nearly 100,000 Claims?
The Similasan eye drop settlement attracted a massive volume of claims for several reasons. The covered products were widely available at major pharmacy chains including CVS and Walgreens, meaning millions of American consumers had purchased them over the seven-and-a-half-year class period. Similasan markets itself as a natural alternative to conventional eye drops, appealing to health-conscious consumers who specifically seek homeopathic products — making the deceptive labeling allegations particularly impactful for this audience.The no-proof-required filing option eliminated the biggest barrier to participation. Most people do not save receipts for $8 to $12 eye drop purchases, so requiring proof would have dramatically reduced the claim rate. By allowing claims under penalty of perjury, the settlement ensured broad participation.
Additionally, word of the settlement spread quickly through social media and class action communities where consumers share information about settlements with easy filing processes and no proof requirements. The growing community of class action enthusiasts who systematically file claims in every available settlement likely contributed significantly to the nearly 100,000 submissions.
Attorneys and Parties in the Similasan Settlement
Class Counsel representing the plaintiff class:• Melissa S. Weiner of Pearson Warshaw LLP
• Rachel Soffin and Nick Suciu of Milberg Coleman Bryson Phillips Grossman PLLC
• Jonas Jacobson of Dovel & Luner LLP
• William H. Anderson of Handley Farah & Anderson PLLC
Defense Counsel: John C. Dougherty, Arameh O’Boyle, and Katherine Galle of Dentons US LLP
Named Plaintiffs: David Plowden, Mario Ortega, and Kamille Faye Vinluan-Jularbal (service awards of up to $2,500 each)
Defendant: Similasan Corporation
Settlement Administrator: Homeopathic Eye Drop Settlement Claims Administrator, 1650 Arch Street, Suite 2210, Philadelphia, PA 19103
What Should You Do Now?
If you received a Similasan eye drop settlement payment via Zelle, Venmo, direct deposit, MasterCard prepaid card, or check, accept or use it. No further action is required. The payment is yours.If you received a paper check, deposit it promptly. Do not let it expire. If you received a MasterCard virtual prepaid card via email, check your inbox and spam folder for an email containing the card details.
Keep records of all payments received for tax purposes. Class action payouts may be taxable, and you may receive a 1099 form if your total payments exceed IRS reporting thresholds. Consult a tax advisor if you have questions.
If you did not file a claim before the October 15, 2025 deadline, you are not eligible to receive a payment from this settlement. The claims period is closed. However, you can sign up for free settlement alerts from OpenClassActions.com to be notified of future class action settlements you may qualify for, including other no-proof-required settlements paying out in 2026.
Similasan Eye Drop Settlement — Frequently Asked Questions
Is the Similasan eye drop settlement payment legitimate?
Yes. The settlement received final approval on July 17, 2025 from a federal judge in Colorado. Payments are being distributed by the court-appointed settlement administrator. If you filed a claim and received a payment, it is real.
How much will I get from the Similasan settlement?
$2.50 per eligible product. Without proof of purchase: up to $10 per household (4 products max). With proof of purchase: $2.50 per product with no cap. Final amounts may be adjusted pro rata depending on total claims.
When are Similasan settlement payments being sent?
Payments are being processed and distributed in early 2026. The settlement received final approval in July 2025 and claims closed in October 2025. Electronic payments arrive faster than paper checks.
Can I still file a claim?
No. The claim deadline was October 15, 2025 and has passed. No new claims are being accepted.
Was proof of purchase required?
No. Consumers could file without receipts for up to 4 products ($10 max per household). Those with proof of purchase could claim every product with no cap.
What products are covered?
16 products: Similasan Dry Eye Relief, Complete Eye Relief, Allergy Eye Relief, Kids Allergy Eye Relief, Red Eye Relief, Pink Eye Relief, Kids Pink Eye Relief, Aging Eye Relief, Computer Eye Relief, Stye Eye Relief, Pink Eye Nighttime Gel, Dry Eye Nighttime Gel, CVS Pink Eye Drops, Walgreens Stye Eye Drops, Walgreens Pink Eye Drops, and Walgreens Allergy Eye Drops.
Are CVS and Walgreens eye drops included?
Yes. CVS Pink Eye Drops, Walgreens Stye Eye Drops, Walgreens Pink Eye Drops, and Walgreens Allergy Eye Drops are all covered. These private-label products were manufactured with ingredients supplied by Similasan.
What is the silver sulfate risk?
The lawsuit alleged Similasan failed to disclose that its eye drops contained silver sulfate, which the FDA does not recognize as safe or effective for OTC eye products. The complaint claimed silver sulfate can decrease night vision and cause irreversible eye and skin discoloration. Similasan denied these allegations.
How many claims were filed?
Nearly 100,000 claim forms were submitted according to court records from the July 17, 2025 final approval hearing.
What payment methods are available?
MasterCard virtual prepaid card, Venmo, direct deposit (ACH), Zelle, or paper check — depending on what you selected when filing.
Will payments be increased or decreased?
Possibly. The settlement allows pro rata adjustments in both directions. If total claims are less than the available fund, payments increase. If claims exceed the fund, payments decrease. The direction depends on final processing results.
Where do leftover funds go?
Any residual funds remaining after all payments are distributed will be donated to The Public Justice Foundation as a cy pres distribution.
Are settlement payments taxable?
Possibly. Class action payouts may be taxable if they exceed IRS reporting thresholds. Consult a tax advisor and keep records of all payments received.
How Do I Find Class Action Settlements?
Find all the latest class actions you can qualify for by getting notified of new lawsuits as soon as they are open to claims:Case Details
Case: Plowden et al. v. Similasan Corporation, Case No. 1:23-cv-02511-DDD-STVCourt: U.S. District Court, District of Colorado
Settlement Amount: $3,575,000
Claim Deadline: October 15, 2025 (passed)
Final Approval: July 17, 2025
Payments: Being distributed (early 2026)
Payment Methods: MasterCard virtual prepaid card, Venmo, direct deposit, Zelle, or paper check
Official Settlement Website: Here
Sources
• OpenClassActions.com — Similasan Eye Drop Class Action Settlement• Settlement Agreement, Case Doc. (November 6, 2024)
• Final Approval Order, Case Doc. (July 17, 2025)
• Official Settlement Website — HomeopathicEyeDropSettlement.com
See full settlement details and claim information on OpenClassActions.com »

DraftKings & FanDuel Addiction Lawsuits
Status: Open
Learn More
Roblox - Investigation
Pre-Qualify Here
Submit Claim
$2.9M Tom's of Maine Toothpaste Settlement
Deadline: July 6, 2026
Submit Claim
$87.5M Beef Prices Settlement
Deadline: June 30, 2026
Submit Claim
Cosequin Dog Supplements Settlement
Deadline: July 21, 2026
Submit Claim
$990K Differin Acne Product Settlement
Deadline: May 19, 2026
Submit Claim
Depo Provera Investigation
Deadline: Pending
Pre-Qualify| Similasan Eye Drop Settlement Summary | |
| Status | Payments Going Out (2026) |
|---|---|
| Settlement Amount | $3,575,000 |
| Payout Per Product | $2.50 (pro rata adjustable) |
| Max Without Proof | $10.00 per household |
| Max With Proof | No cap |
| Claims Filed | ~100,000 |
| Products Covered | 16 (Similasan, CVS, Walgreens) |
| Payment Methods | Prepaid Card, Venmo, Direct Deposit, Zelle, Check |
| Claim Deadline | October 15, 2025 (passed) |
| Final Approval | July 17, 2025 |
| Case | Plowden v. Similasan Corp., No. 1:23-cv-02511 |
| Court | D. Colo. |